Predictive Modeling of Emerging Tropical Disease
The burden of emerging infectious diseases and neglected tropical diseases is high in Central Africa’s Congo Basin, fueled by endemic viral and parasitic diseases as well as low public spending on health care.
CBI researchers have expertise in wildlife sampling, ecological modeling, and laboratory assays to detect tropical diseases, and are applying this expertise to predict present-day and future distributions of emerging infectious diseases and neglected tropical diseases.
Several of these diseases, including monkeypox and Ebola, can be transmitted to humans through contact with wildlife. Understanding the spatial and temporal patterns of NTD outbreaks can provide information – in near real-time – to public health decision-makers, helping inform disease control policies.
Although other research groups have mapped the risk of NTDs, the spatial resolution of these efforts has been limited to the county or district scale. In contrast, CBI researchers have experience mapping disease risk at the continental scale – across all of Africa – which can help determine areas of the greatest risk, thus helping policy-makers allocate treatments more effectively. CBI research staff are working on Ebola, monkeypox, avian and swine influenza, Zika, and malaria.
Ebola
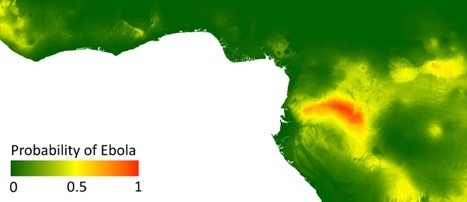
At least 25 separate outbreaks of the Ebola virus have been recorded in West and Central Africa since 1976, although none was more devastating than the 2014 outbreak, which primarily occurred in Guinea, Sierra Leone, and Liberia. CBI has amassed a dataset of nearly 400 individual locations and case counts for this outbreak, and is applying non-parametric techniques – normally used to forecast earthquakes and wildfires – to these data to predict how the disease spread among regions. These models perform remarkably well, suggesting they can maximize efficiency in control efforts while outbreaks are occurring, rather than after they have already had devastating effects. CBI researchers have also developed ecological niche models, using Maxent, in collaboration with national experts in Cameroon.
Monkeypox
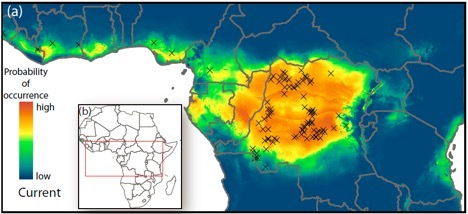
Monkeypox is a zoonotic virus considered the most important orthopoxvirus in humans since the eradication of its close relative, smallpox. Smallpox immunization is thought to have conferred immunity to monkeypox: Since the cessation of smallpox vaccination in the 1980s, there has been a 20-fold increase in the number of human monkeypox cases reported in the Democratic Republic of Congo. CBI researchers have used occurrence of these diseases to identify several likely reservoir species for this virus, including multiple members of the rope squirrel family (Genus Funiscurus). Researchers have also determined several environmental factors associated with higher risk of exposure to monkeypox, such as proximity to dense forests, and habitats suitable for the rope squirrel reservoirs.
Monkeypox is a DNA virus, so it can be detected in host tissues for a prolonged period: CBI scientists have detected monkeypox in centuries-old mammal museum specimens from Central Africa. With cutting-edge DNA extraction methods and modeling techniques, CBI researchers can uncover how the spread of disease has changed over time in the Congo Basin.
Avian and swine influenza
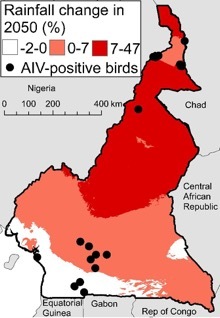
In 2006, avian influenza was detected in arid areas in northern Cameroon. Climate change is predicted to dry these areas even further drier over the next four decades. This could cause crowding of wild birds around the remaining water bodies, increasing opportunities for influenza transmission. Researchers at CBI are working to characterize the spatial pattern of influenza prevalence in Central Africa, and identify the most important host species. They have screened thousands of samples from wild and domestic birds, as well as pigs, and already uncovered some important insights.
Researchers found the prevalence of avian influenza in songbirds is equivalent to that of waterfowl and shorebirds, long considered the only important wild bird reservoirs of influenza. CBI research has also shown that the swine influenza virus, which emerged in Mexico and caused a human pandemic in 2009, spread to Central Africa the following year. The virus was transmitted from humans back to pigs, infecting up to 80 percent of herds.
Zika
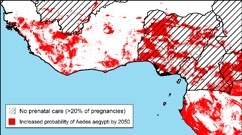
The Zika virus – discovered in Uganda in 1947 – is an arbovirus whose transmission occurs primarily via mosquito vectors, though it is also sexually transmitted. In 2014, Zika infected thousands of people when it spread to the Western hemisphere. In women infected during pregnancy, more than half of all Zika infections result in abnormal fetal outcomes, including microcephaly. Approximately 1 percent of non-pregnant adults infected with Zika develop Guillain-Barré Syndrome, a form of paralysis. Researchers at CBI are modeling the risk of Zika in Africa and predicting how climate change will impact the distribution of its mosquito vectors.
Avian blood parasites
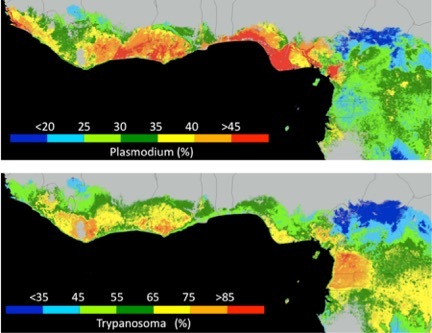
CBI researchers are exploring the geographic distributions and ecological requirements of avian blood parasites (Plasmodium and Trypanosomes) in Central Africa. This research has led to a variety of interesting discoveries, including new competent vectors of malaria as well as ecological conditions driving high malaria prevalence rates in Central Africa. More recent work has focused on identifying vector/host systems and the mechanisms of host shifts, and estimating the impacts of deforestation on the presence and prevalence of various malaria parasite strains. As Central Africa faces some of the highest land use changes and deforestation rates anywhere in the world, understanding how these processes affect the natural ecology of infectious diseases will be critical to inform public health decisions under these new conditions.